 Horaizon Orthopedic Implants
Horaizon Orthopedic Implants
 Horaizon Orthopedic Implants
Horaizon Orthopedic Implants
Precision-engineered orthopedic medical devices — combining advanced R&D, world-class manufacturing, and a commitment to advancing human health worldwide.

Zhejiang Horaizon Medical Device Co., Ltd. is a fully integrated company combining development, manufacture, and sales, specializing in orthopedic medical devices. Founded with a clear mission to advance surgical outcomes, Horaizon has grown into a globally recognized name trusted by surgeons, hospitals, and distributors across six continents.
Our company possesses advanced high-precision, large-scale production equipment and complete sets of precision testing instruments. A dedicated professional team drives technical development, ensuring every product meets the most rigorous international standards before reaching the operating room.
Company management is based on ISO 9000 / ISO 13485 International Quality System Standards and the YY/T0287 Medical Device Quality System Standard — reflecting our unwavering commitment to product safety, traceability, and continuous improvement.

Every product, every process, and every partnership is guided by three foundational principles that define who we are and how we operate.
From raw material sourcing to final inspection, precision is embedded in every stage of our manufacturing process. Our CNC machining centers and CMM measurement systems achieve tolerances within ±0.01mm, ensuring implants and instruments perform exactly as designed in critical surgical environments.
Our engineering and R&D team holds deep expertise in biomechanics, materials science, and surgical technique. We collaborate closely with orthopedic surgeons and clinical specialists to develop products that solve real-world challenges — from complex spinal reconstruction to minimally invasive joint replacement.
Innovation drives our product roadmap. We continuously invest in next-generation implant designs, biocompatible surface coatings, and smart instrument systems. Our R&D center dedicates over 8% of annual revenue to new product development, keeping Horaizon at the forefront of orthopedic technology.
Our state-of-the-art production facility in Zhejiang, China spans over 15,000 m² and is equipped with the latest precision manufacturing and quality control technology.
Over 80 five-axis CNC machining centers enable high-volume, high-precision production of titanium, stainless steel, and PEEK implants and instruments with consistent repeatability.
Complete sets of precision testing equipment including CMM coordinate measuring machines, surface roughness testers, fatigue testing systems, and biocompatibility analysis tools ensure every product meets specification.
ISO Class 7 clean rooms dedicated to final assembly and packaging of sterile orthopedic implants, fully compliant with EU MDR and FDA 21 CFR Part 820 environmental requirements.
In-house anodizing, sandblasting, HA coating, and titanium nitride PVD coating capabilities allow full control over implant surface properties for optimal osseointegration and wear resistance.
EO sterilization partnerships and validated packaging lines ensure products are delivered shelf-ready with full traceability documentation, UDI compliance, and multi-language labeling support.
Lean production systems and digital MES (Manufacturing Execution System) integration enable real-time production tracking, rapid changeover, and on-time delivery rates exceeding 98%.
Surgeons and distributors worldwide choose Horaizon not just for our products — but for the complete partnership, support, and reliability we deliver at every stage.
Our regulatory affairs team has successfully obtained CE Mark, FDA 510(k) clearance, and NMPA registrations for multiple product lines, providing partners with a clear pathway to global market access.
Every distributor receives a dedicated account manager, comprehensive product training, surgical technique guides, and marketing materials — ensuring you have everything needed to succeed in your market.
With robust inventory management and streamlined logistics, standard orders ship within 7–14 business days. Consignment set programs and loaner instrument systems are available for key accounts.
Our vertically integrated manufacturing model eliminates middlemen, allowing us to offer premium-quality orthopedic devices at highly competitive price points — maximizing your margin without compromising patient outcomes.
Need a modified implant geometry or a custom instrument set? Our engineering team can develop tailored solutions with full documentation, design validation, and regulatory support included.
Horaizon products are distributed in over 60 countries through a network of trusted regional partners. Our global logistics infrastructure ensures reliable supply chains regardless of your location.
Our quality management system and product registrations demonstrate our commitment to the highest standards of safety, efficacy, and regulatory compliance.
| Certification / Standard | Scope | Issuing Body | Status |
|---|---|---|---|
| ISO 13485:2016 | Medical Device Quality Management System | TÜV / BSI | ✓ Active |
| ISO 9001:2015 | General Quality Management System | Accredited CB | ✓ Active |
| CE Mark (MDR) | Orthopedic Implants & Instruments — EU Market | Notified Body | ✓ Active |
| FDA 510(k) | Selected Orthopedic Device Lines — US Market | U.S. FDA | ✓ Active |
| NMPA Registration | Orthopedic Medical Devices — China Market | NMPA China | ✓ Active |
| YY/T 0287 | Medical Device Quality System Standard | SAMR China | ✓ Active |
Every Horaizon product passes through a rigorous five-stage development and manufacturing process designed to ensure clinical reliability and patient safety.
Surgeon collaboration, biomechanical analysis, and FEA simulation to validate design intent.
Rapid prototyping, cadaveric lab testing, and design verification against performance specifications.
CNC machining, surface treatment, and clean-room assembly with in-process quality checks.
100% dimensional inspection, fatigue testing, biocompatibility verification, and final QC release.
Regulatory filing, validated packaging, sterilization, UDI labeling, and on-time worldwide delivery.
From large hospital systems to independent distributors, Horaizon has delivered proven orthopedic solutions across diverse clinical settings and regulatory environments.
Supplied a complete pedicle screw and rod spinal fusion system to a 12-hospital group in Germany, including custom instrument sets and comprehensive surgical training. Over 800 cases performed with a reported complication rate below 1.2%.
Spine CE Mark Custom InstrumentsEstablished a regional distribution partnership for our cementless hip stem and cup system across Brazil and Colombia, achieving ANVISA and INVIMA registration. Annual volume exceeds 2,000 implants with growing market share.
Hip ANVISA OEMLong-term OEM manufacturing agreement for a full line of trauma fixation plates and screws under a US distributor's private label. FDA 510(k) clearance obtained, with annual production exceeding 50,000 units and 99.6% on-time delivery rate.
Trauma FDA 510(k) Private LabelHoraizon is an experienced OEM partner for orthopedic companies worldwide, offering full-service private-label manufacturing with regulatory support from concept to commercialization.
Horaizon has served as the manufacturing backbone for orthopedic brands across North America, Europe, Australia, and Southeast Asia. Our OEM clients benefit from our deep regulatory expertise, established supply chains, and a quality system that has passed multiple third-party audits.
Whether you are launching a new orthopedic brand, expanding your product portfolio, or seeking a more reliable manufacturing source, Horaizon offers the capabilities, certifications, and commitment to make your program a success.
Minimum Order Quantities: Flexible — starting from 50 units per SKU for pilot programs.
Lead Time: Standard products 4–8 weeks; custom products 12–20 weeks including tooling.
Sample Policy: Engineering samples available for evaluation prior to full commitment.
Trusted by surgeons, distributors, and healthcare institutions worldwide — here is what our partners have to say about working with Horaizon Medical.
Horaizon's spinal fusion system has become our go-to choice for complex reconstructions. The instrumentation is intuitive, the implants are precisely machined, and their technical support team is always responsive. Exceptional quality at a competitive price point.
We have been distributing Horaizon trauma products in Southeast Asia for over five years. Their on-time delivery, consistent quality, and willingness to support our local regulatory filings have made them our most valued manufacturing partner.
The OEM program with Horaizon has been transformative for our business. They handled the FDA documentation, produced flawless private-label products, and delivered on schedule every single time. I would recommend them to any orthopedic company looking for a reliable OEM partner.
Whether you are a surgeon seeking reliable orthopedic implants, a distributor expanding your portfolio, or a company looking for an experienced OEM manufacturing partner — Horaizon is ready to support your success.
Contact Us Today →



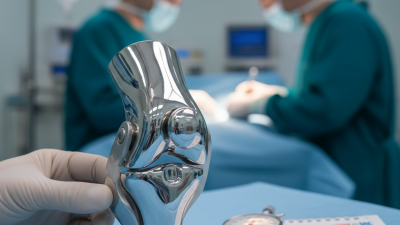
The demand for custom orthopedic implants has surged in recent years. With advancements in technology, these implants are tailored to fit individual
Author: Amelia Date: 2026-03-30
Learn More
The 2026 Canton Fair presents a unique opportunity for sourcing Wrist External Fixators. As we enter a new era of healthcare solutions, industry
Author: Madeline Date: 2026-03-27
Learn More
The Canton Fair 2026 provides an excellent opportunity for sourcing Tibial Intramedullary Nails. Taking place from April 15 to May 5, this event
Author: Charlotte Date: 2026-03-25
Learn More