 Horaizon Orthopedic Implants
Horaizon Orthopedic Implants
 Horaizon Orthopedic Implants
Horaizon Orthopedic Implants

Surgical Dtn Retrograde Tibia Nail Instrument - New York Edition

Medical Orthopedic Spine Endoscopy Instrument Set Surgery Used - Houston Series

Sterilized Package Prosthesis Knee Joint - Chicago Industrial Grade

CE Certified Orthopedic Pfna Instrument Set Essential Bone Instruments - California Standard
In the United States, the demand for Revision Hip and Knee Systems has seen an unprecedented surge. As the "Baby Boomer" generation remains active longer into their senior years, the lifespan of primary orthopedic implants is being pushed to the limit. Our role as a premier manufacturer and factory for the United States market is to provide secondary surgical solutions that address bone loss, infection, and implant instability.
The industrial landscape in the US for orthopedic devices is characterized by rigorous FDA oversight and a shift toward Value-Based Healthcare. Surgeons in top-tier institutions like the Mayo Clinic or Johns Hopkins now demand revision systems that offer modularity and long-term biological fixation. Our factory integrates high-precision CNC machining with additive manufacturing to create components that mirror the anatomical needs of the diverse US patient population.

Annual Revision Procedures in USA
Expected Annual Growth Rate (CAGR)
Biocompatibility Success Rate
Global Quality Standard Compliance
Using artificial intelligence, we analyze thousands of CT scans from North American patients to optimize the taper and stem length of our revision hip systems. This ensures a "first-fit" accuracy that reduces time in the operating room.
Our US-bound products utilize world-leading 3D printing techniques to create highly porous titanium structures. These structures encourage rapid bone ingrowth, a critical factor for successful revision surgery where bone stock is often compromised.
Ultra-High-Molecular-Weight Polyethylene (UHMWPE) inserts are engineered for maximum wear resistance. In the US, where patients live longer, our highly cross-linked polyethylene significantly reduces osteolysis risks.
Zhejiang Horaizon Medical Device Co., Ltd. is a company combined with development, manufacture, and sales, and is specialized in orthopedic medical devices. Our company possesses advanced production equipment with high-precision and large-scale and complete sets of precision testing equipment.
We have a professional team responsible for technical development. The company management is based on ISO9000 / ISO 13485 International Quality System Standard and YY/T0287 Medical Device Quality System Standard. We are always in pursuit of more stable and advanced product technology, and more convenient and simpler operational function.
Horaizon people adhere to the operation concept in mind, which is "precision, professional, innovative". We will strive to make new value for the community and keep forging ahead for human health and happiness through excellent products with best quality and outstanding service!

In major US industrial hubs—from the medical device corridor in Indiana to the biotech labs in Massachusetts—the trend is moving toward Modular Revision Systems. Unlike primary arthroplasty, revision surgery often requires the surgeon to "build" the implant based on the specific deficit found during surgery. Our factory focuses on producing interchangeable stems, sleeves, and augments that allow US surgeons the flexibility to address complex femoral and acetabular defects.
Furthermore, the integration of Robotic-Assisted Surgery in United States hospitals has revolutionized how our systems are applied. Precision robotic arms require implants with tight tolerances and specific digital footprints. As a leading manufacturer, we ensure our revision knee and hip components are compatible with the latest robotic navigation software used in American operating theaters.
In the high-pressure environment of US surgical centers, efficiency is paramount. Our instrument sets, such as the PFNA Instrument Set and the Expert Femoral Instrument Set, are designed for rapid sterilization and intuitive layout, minimizing "down-time" between surgeries in busy urban hospitals in cities like Miami, Dallas, and Seattle.

Prosthesis Replacement Orthopedic Sterilized Package Class III Knee Joint
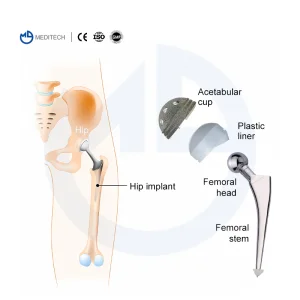
Hip Artificial Knee Joint Orthopedic Implants Cemented Stem Hip Replacement

CE Certified Orthopedic Expert Femoral Instrument Set for Shoulder Surgery

S-ROM Titanium Alloy Cementless Hip Joints for Revision Arthroplasty

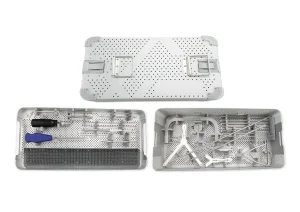
Orthopedic Surgery Osteotomy Instrument Set Trauma Instrument

Orthopedic Surgical Instruments Box Aluminum Sterilization Container

Replacement Sterilized Package Prosthesis Knee Joint World′ S Leading 3D Printed
Whether you are a hospital procurement officer in New York or an orthopedic surgeon in Los Angeles, our systems provide the reliability and innovation required for the most complex revision cases. Contact us today for technical specifications or volume manufacturing partnerships.
Send Inquiry Now