1 / 5
 Horaizon Orthopedic Implants
Horaizon Orthopedic Implants










| Still deciding? Get samples of $ ! US$ 80/Piece Request Sample |
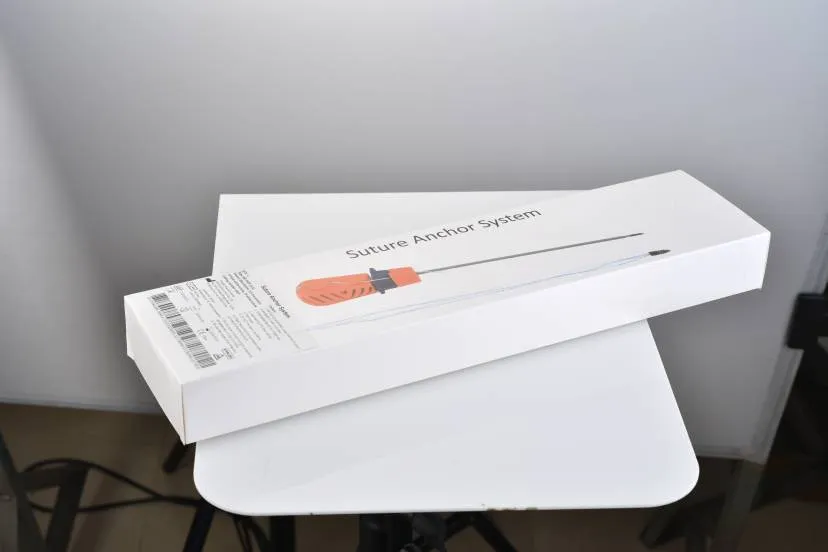
Suture Anchor Material Arthroscopy Gravity Peek Standard Anchors
| Product Name | PEEK Suture Anchor System |
|---|---|
| Ctn Size | 42 * 22.5 * 25.5 CM |
| G.W./CTN | 1.5 KG - 1.8 KG |
| Certificates | CE & ISO13485 |
| Validity Period | 3 years |

Suture Anchor System consists of anchor, nonabsorbable suture and inserter. Anchor is made of PEEK. Suture is made of ultra high molecular weight polyethylene (UHMWPE) material without coating, woven from several UHMWPE yarns meeting ASTM F2848-17 requirements. The blue suture is dyed with FDA-approved material (code 73.1015). The inserter part contacting the human body is made of stainless steel conforming to ASTM F899-12b, while the handle is made of polycarbonate and ABS material.