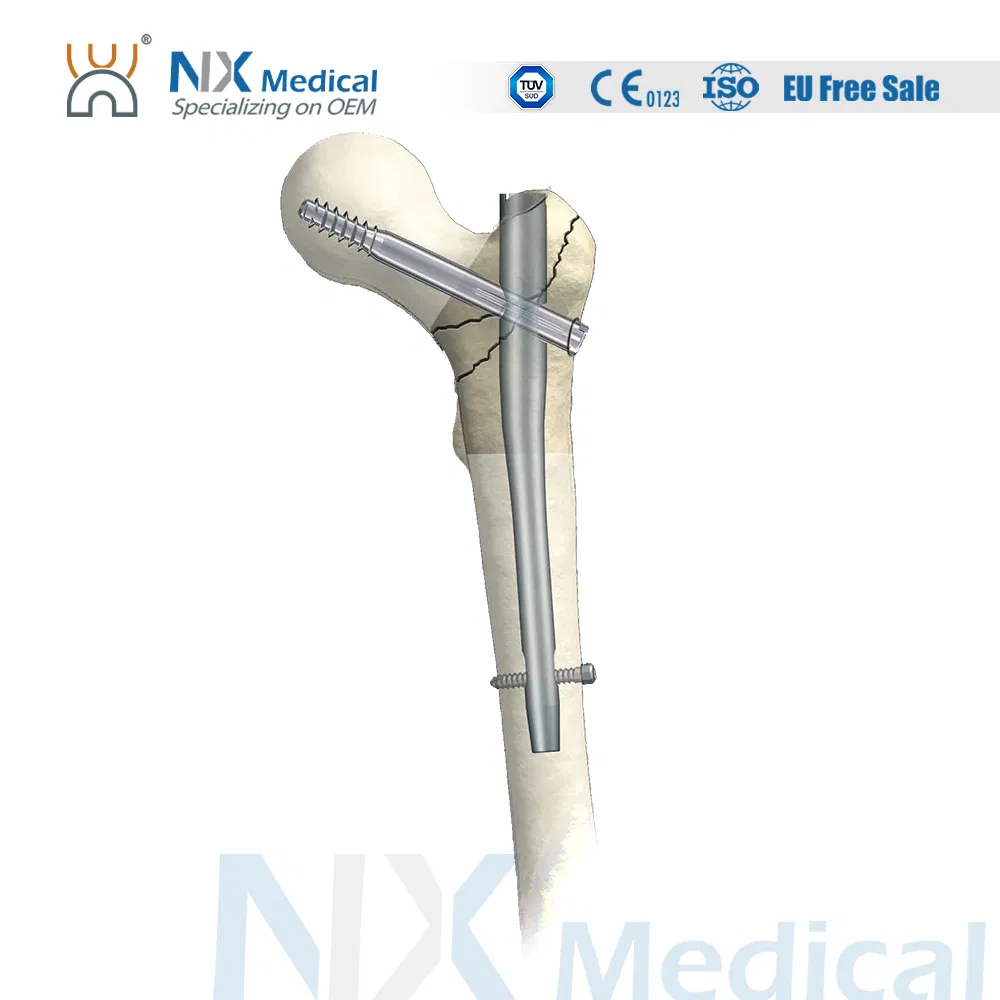
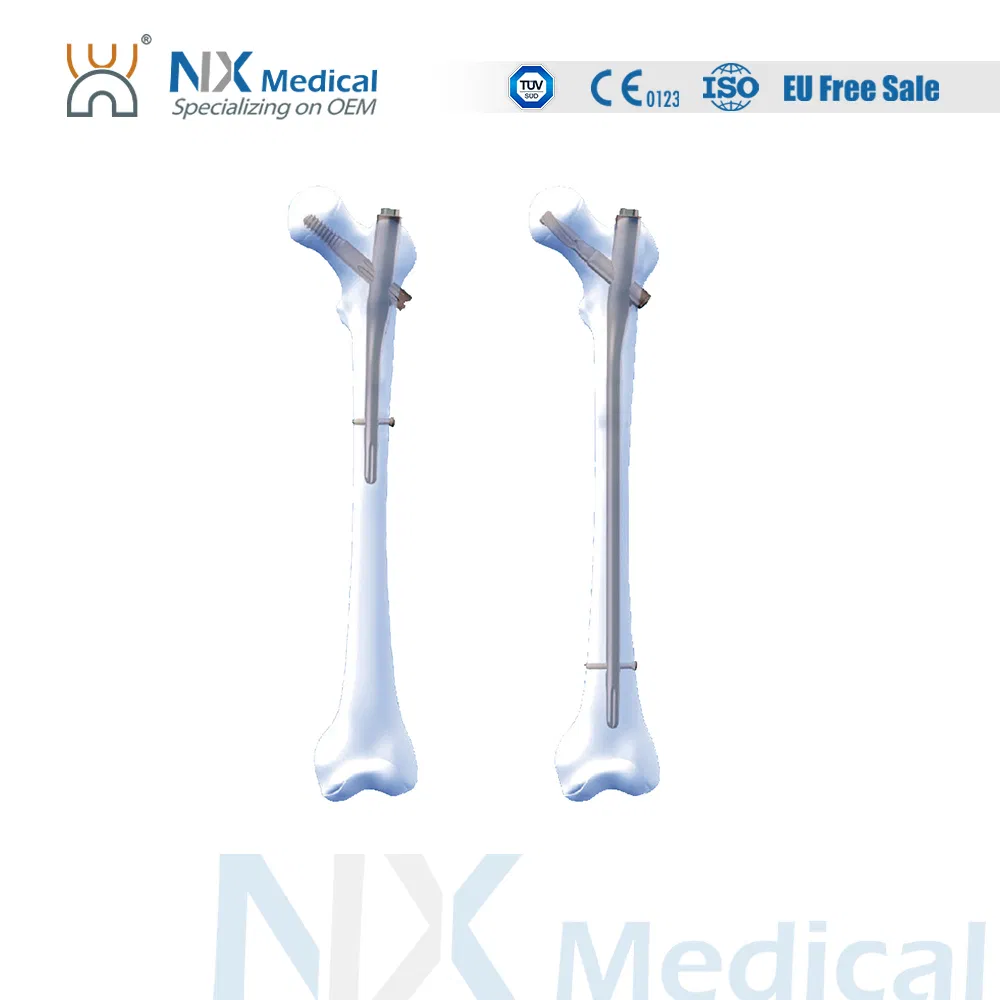



 Horaizon Orthopedic Implants
Horaizon Orthopedic Implants










| Still deciding? Get samples of $ ! US$ 80/Piece Request Sample |

Key Features and Benefits:
| Product Description | Size (mm) | Direction | Material |
|---|---|---|---|
| Gamma C Nail (125°) | 9 x 180 / 200 / 220 / 240 | Universal | Titanium Alloy |
| 9 x 320 / 340 / 360 / 380 / 400 | Left / Right | Titanium Alloy | |
| 10 x 180 / 200 / 220 / 240 | Universal | Titanium Alloy | |
| 11 x 320 / 340 / 360 / 380 / 400 / 420 | Left / Right | Titanium Alloy | |
| Gamma C Nail (130°) | 12 x 180 / 200 / 220 / 240 | Universal | Titanium Alloy |
| 13 x 340 / 360 / 380 / 400 / 420 / 440 | Left / Right | Titanium Alloy |
Our company is a leading manufacturer specializing in orthopedic medical devices. We provide comprehensive, cost-effective, and reliable therapeutic solutions to surgeons and patients worldwide. Our extensive portfolio includes high-quality trauma implants, spinal systems, and precision surgical instruments.
Committed to innovation, we invest heavily in R&D to ensure our products meet the highest standards of safety and efficacy. Our manufacturing processes are strictly compliant with ISO13485, and our products carry TUV CE0123 and NMPA certifications.
Our goal is to provide advanced orthopedic implants and technologies that ensure reliable procedures, rapid recovery, and a pain-free life for patients after surgery. We also serve as a major OEM partner for large global orthopedic companies, maintaining a reputation for excellence in the international market.



