

 Horaizon Orthopedic Implants
Horaizon Orthopedic Implants










| Still deciding? Get samples of $ ! US$ 100/Set Request Sample |


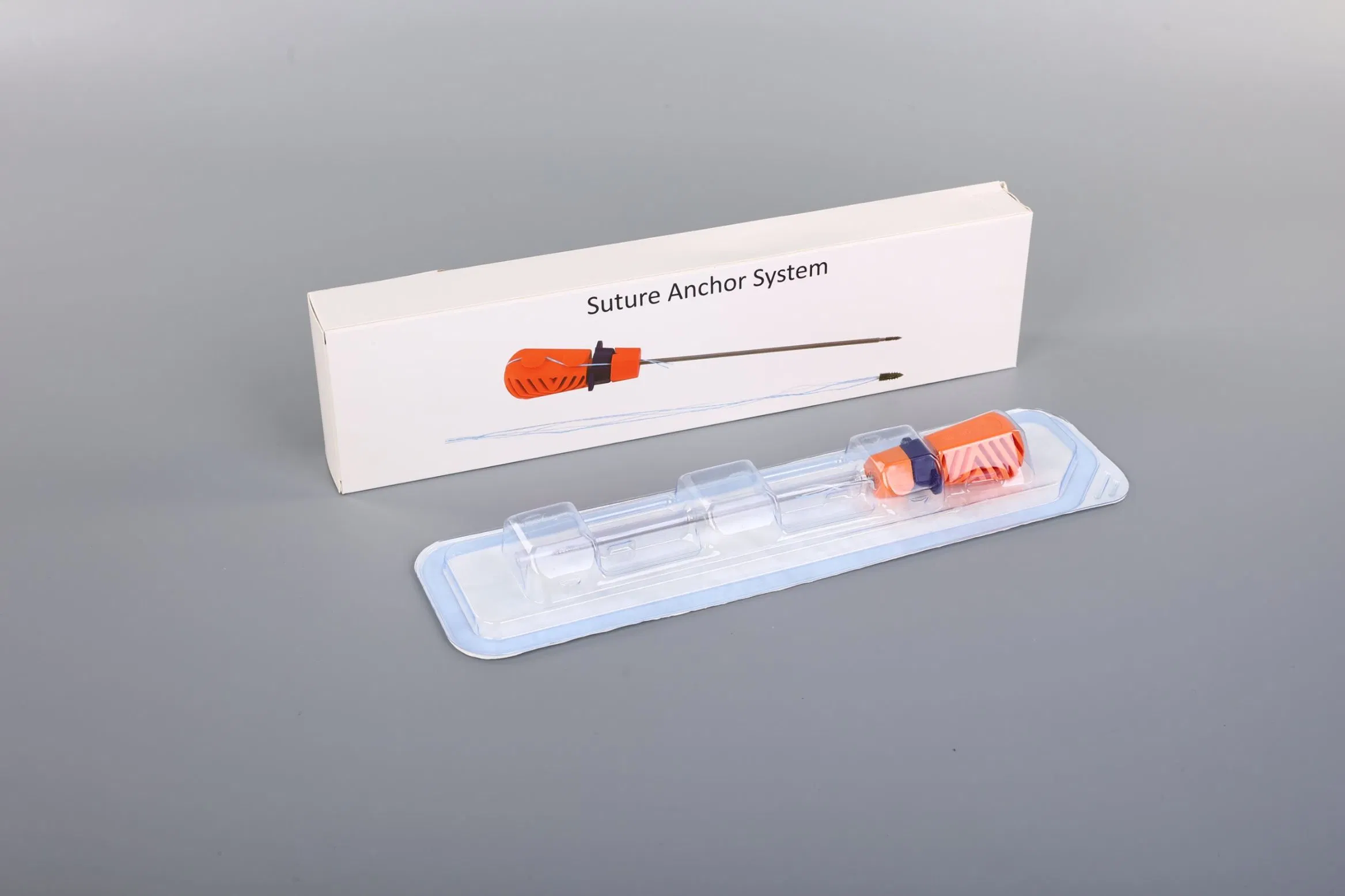
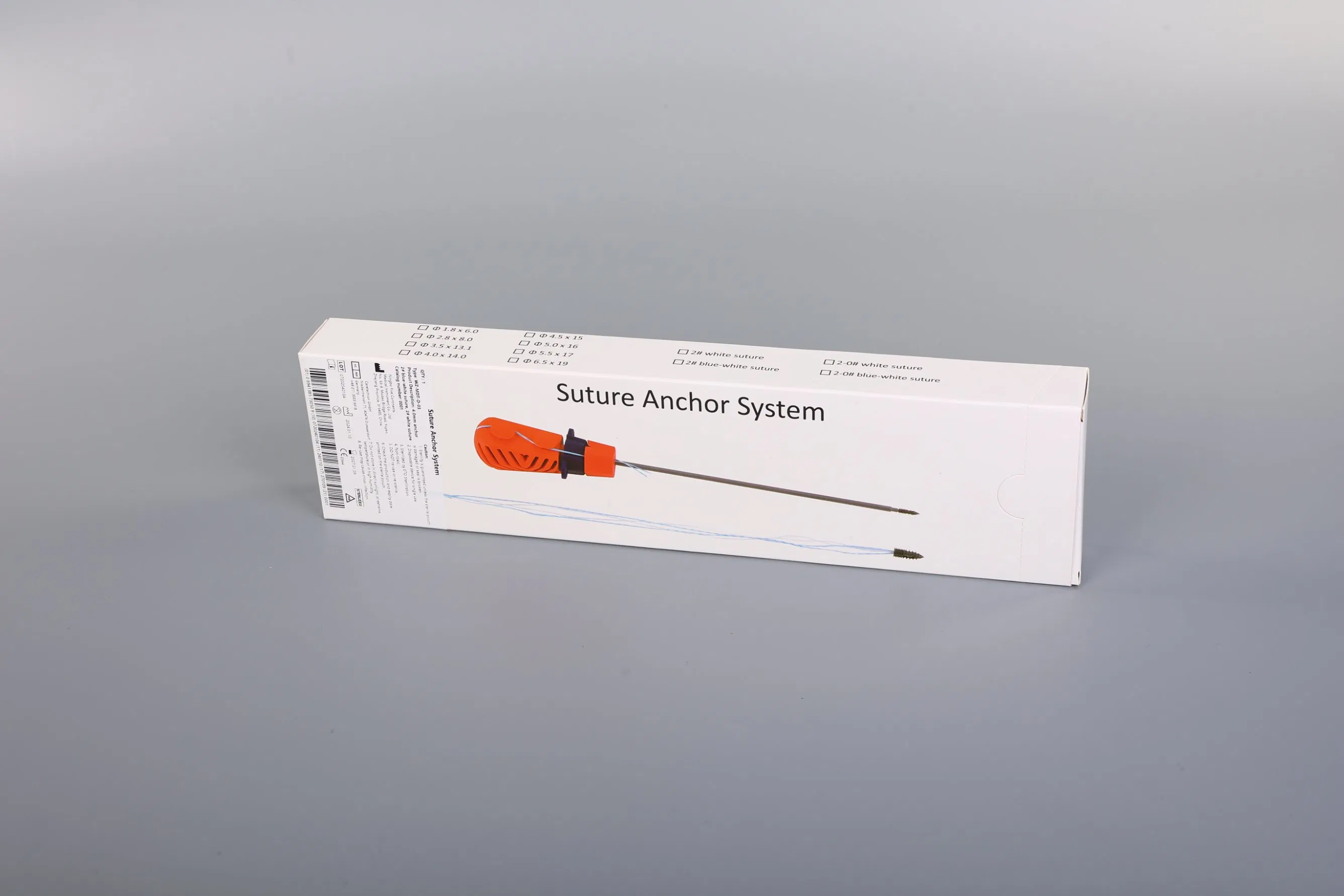
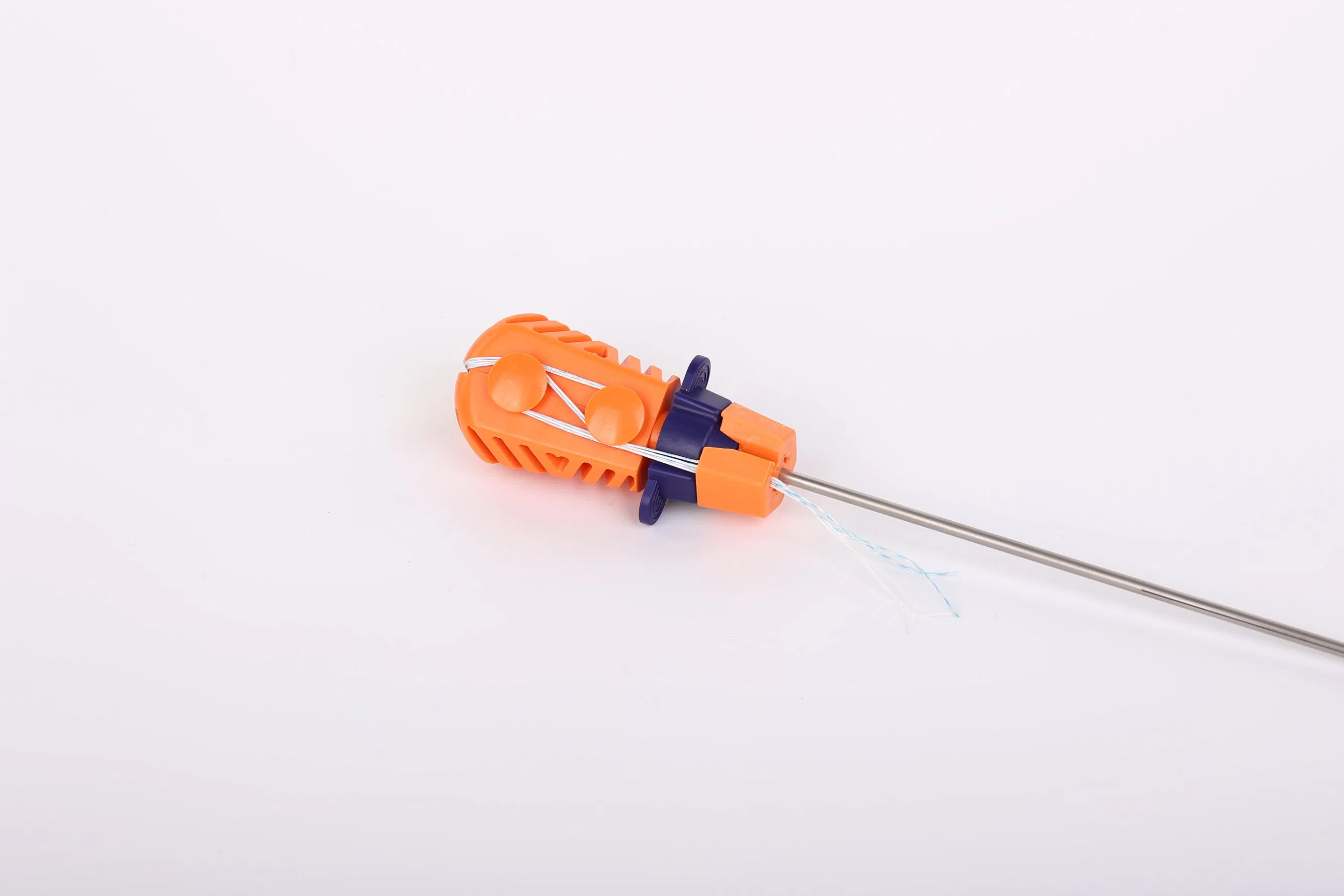
Suture Anchor System: This advanced medical system consists of an anchor, nonabsorbable suture, and a specialized inserter. The anchor is precision-crafted from Ti6Al4V alloy, strictly adhering to ISO5832-3:2016 requirements, ensuring biocompatibility and strength for orthopedic applications.
Suture Technology: The system utilizes sutures made of ultra-high molecular weight polyethylene (UHMWPE) material. These sutures are woven from multiple UHMWPE yarns (meeting ASTM F2848-17 standards) without additional coating. The distinctive blue suture is dyed using FDA-approved materials, ensuring safety and high visibility during surgical procedures.
Instrument Design: The inserter is designed for ergonomic precision. Components contacting the human body are constructed from medical-grade stainless steel (conforming to ASTM F899-12b). The handle, designed for a secure non-slip grip, is manufactured from durable polycarbonate and ABS materials.

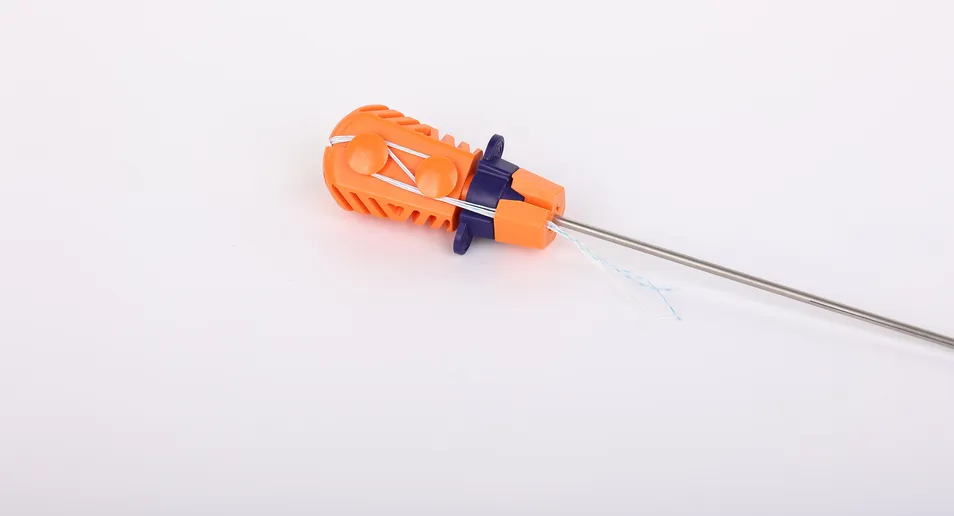

A comprehensive range of orthopedic surgical consumables is available, including:
All products are fully compliant with CE certification and ISO 13485 standards, ensuring reliable performance for medical institutions globally.


