




 Horaizon Orthopedic Implants
Horaizon Orthopedic Implants










| Customization: | Available |
|---|---|
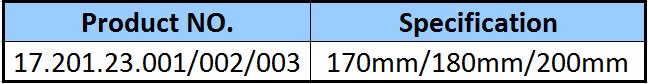
| Type: | Surgical Needle & Hook |
| Application: | Orthopedic |
As a national high-tech enterprise, R&D investment consistently exceeds 8% of total sales. This focus ensures the transformation of clinical expertise into innovative medical solutions, resulting in numerous national invention patents and utility model patents.
The facility pioneers global revolutionary technologies including the Rev Drill system, Three-Row-Stapler, Sternal Fixation, and Rib plate, which are widely endorsed by clinical experts and the international market.

"Good faith as the base, quality above all" is the core philosophy. Operations strictly adhere to the ISO13485 quality system and CE MDD 93/42/EEC directives.
The production line features high-end international equipment, including:
Quality assurance is maintained through three critical points: raw material procurement control, product quality control, and new product performance analysis.

Medical products are exported globally to countries including Columbia, France, Turkey, Indonesia, Mexico, Russia, and Thailand. Feedback from these markets confirms that product quality consistently meets local and international regulatory standards.



The enterprise serves as a major OEM supplier to some of the largest global players in the orthopedic and medical device industry, providing customized manufacturing solutions with high precision.