



 Horaizon Orthopedic Implants
Horaizon Orthopedic Implants










| Still deciding? Get samples of $ ! US$ 20.3/Piece Request Sample |

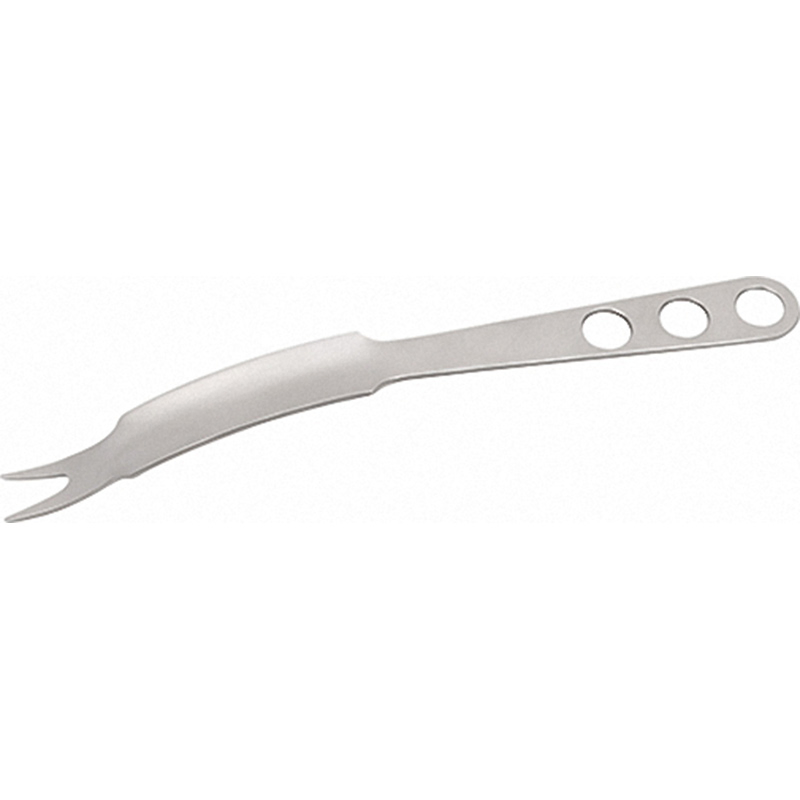
| Product Name | Double Head Hook |
| Model Number | 686 |
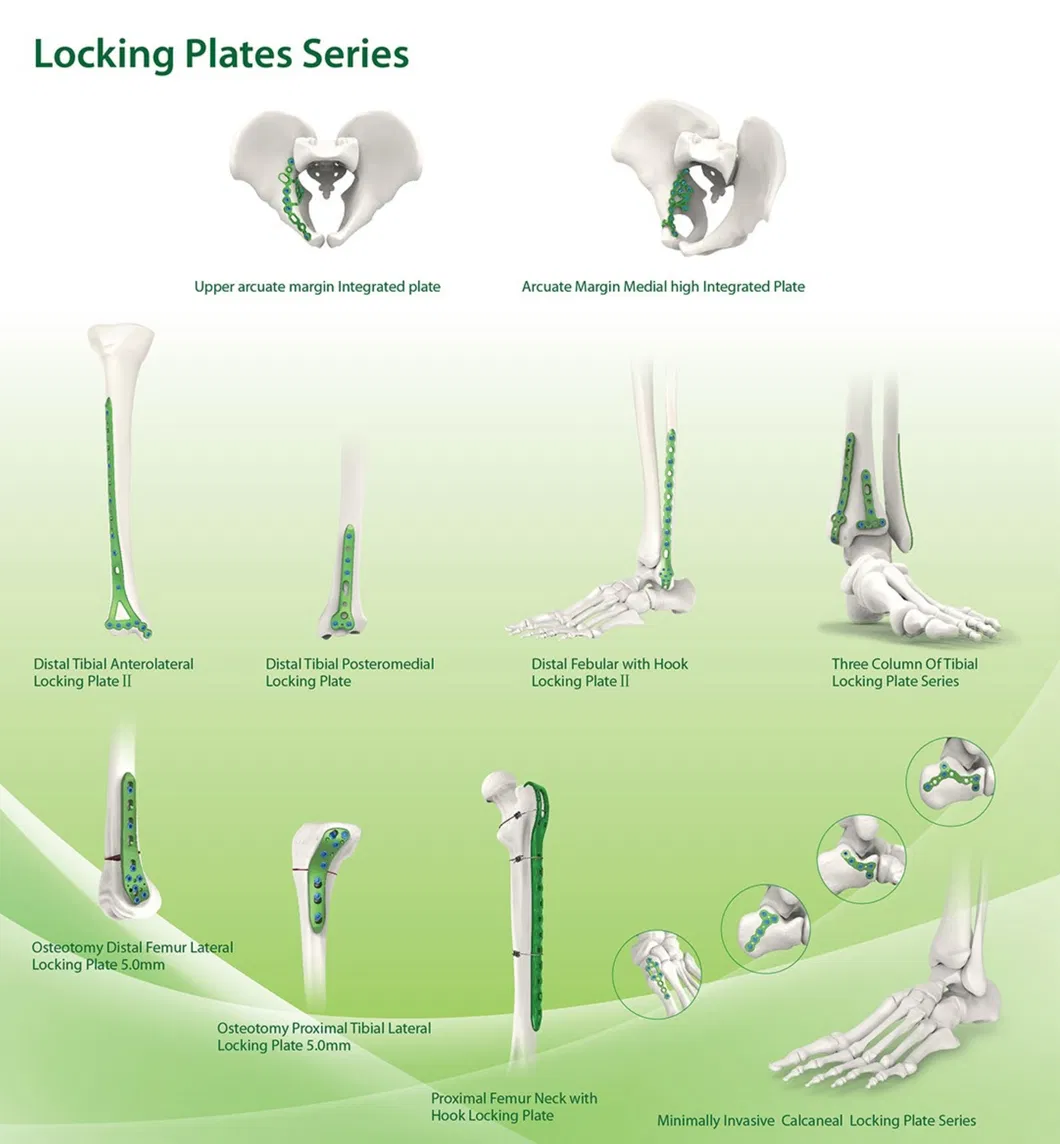
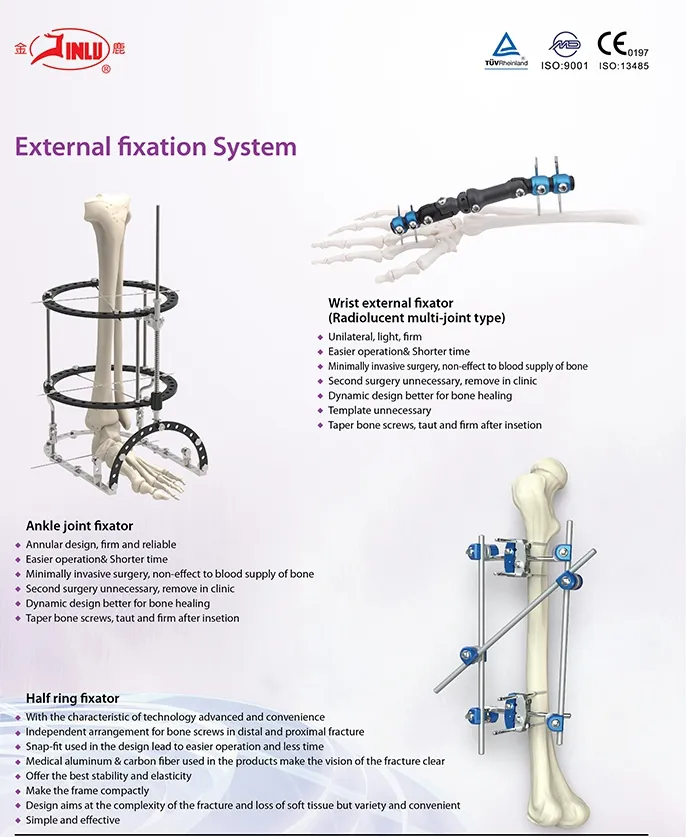






Established in 1958, our company is dedicated to the development, manufacture, and sales of orthopedic implants and instruments. As a key enterprise for national medical apparatus, the product line covers Spine pedical screws, Interlocking nails, Locking plates/screws, trauma plates/screws, cannulated screws, external fixators, hip prosthesis, and surgical instruments.
All medical apparatus have passed the approval and authentication of professional superintendent offices. Products are characterized by multi-varieties and complete specifications, featuring laser anti-fake marks and quality responsibility insurance.
Advanced equipment, including imported CNC and high precision digital control machines, combined with a 100,000 Grade axenic purification chamber, contribute to high-quality standards. We are among the first to pass ISO 9001 quality system certification and have received numerous science and technology awards.
