


1 / 5
 Horaizon Orthopedic Implants
Horaizon Orthopedic Implants










| Still deciding? Get samples of $ ! US$ 100/Set Request Sample |
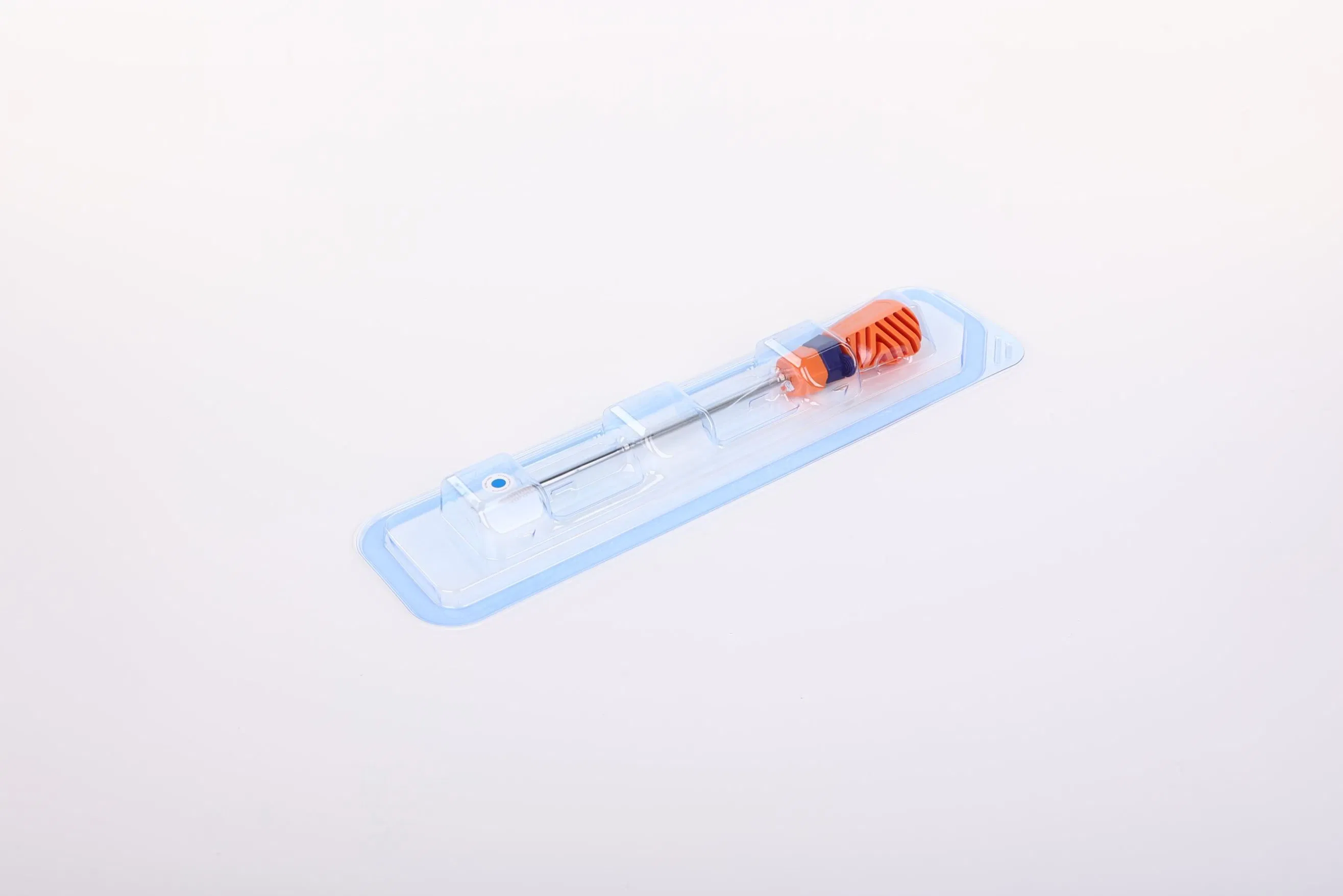
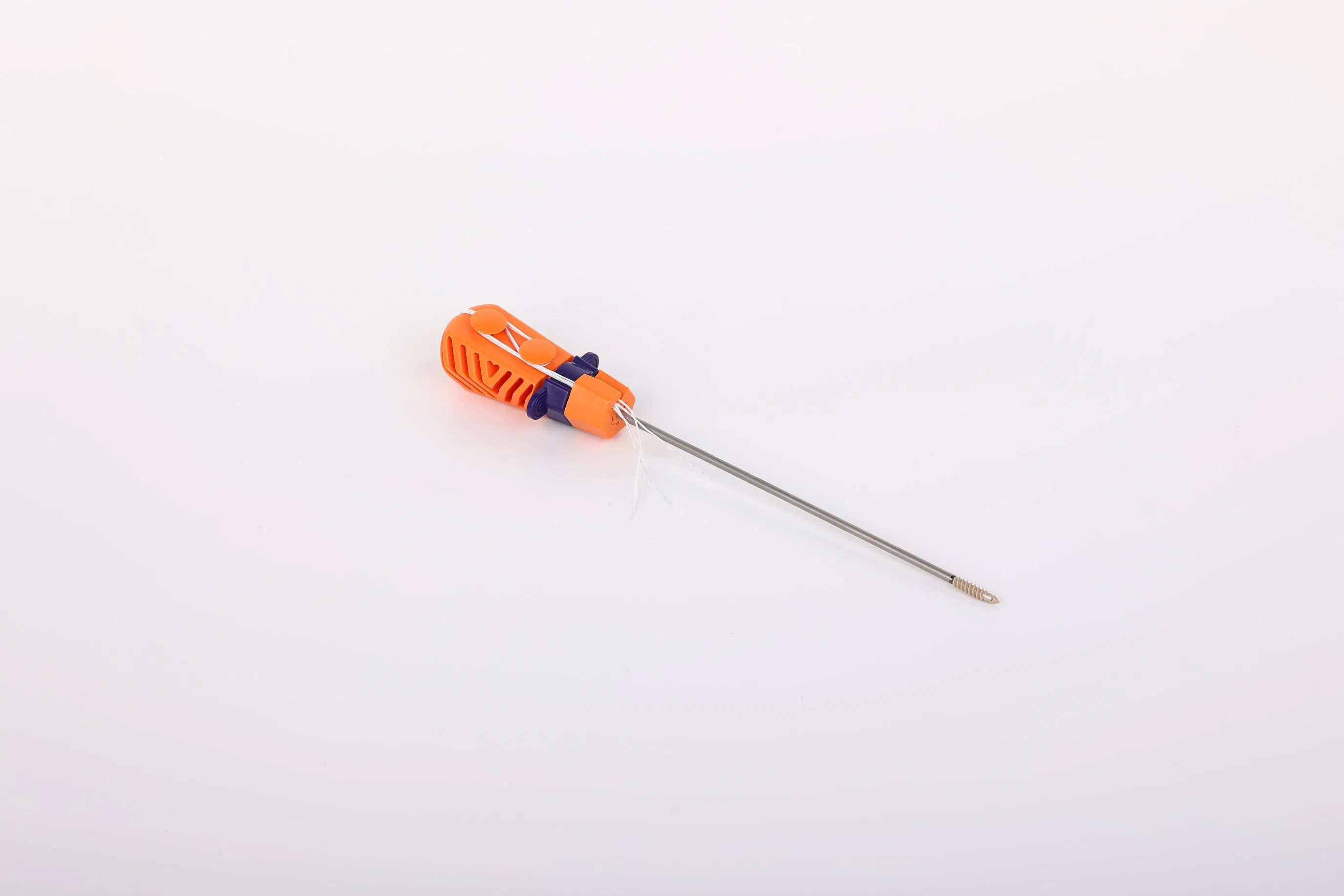
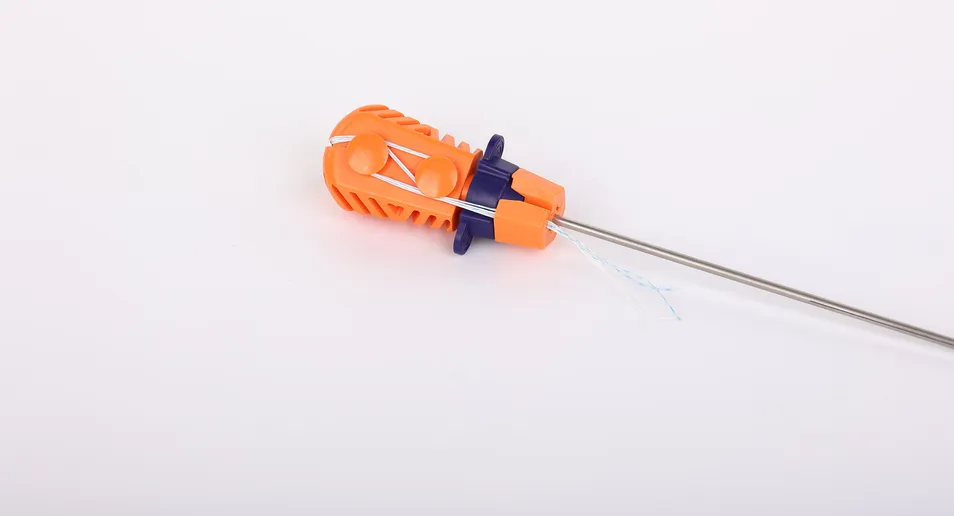
Factory Direct Price Multiple Specifications Suture Anchor Bolt with Line for Joint Repair Surgery



The Suture Anchor System consists of an anchor, nonabsorbable suture, and inserter.

The production process utilizes a Class 100,000 clean workshop and a Class 10,000 purification laboratory to ensure product quality and safety. Our comprehensive range includes orthopedic surgical consumables such as Disposable Surgical Lavage Systems, Meniscal Repair Systems, Shaver Blades, and Bone Cement Mixers. All products adhere to international quality benchmarks, including CE certification and ISO 13485 standards.


