




1 / 5
 Horaizon Orthopedic Implants
Horaizon Orthopedic Implants










| Customization: | Available |
|---|---|
| Certification: | ISO, CE |
| Disinfection: | Disinfection |
Basic Information
Product Description
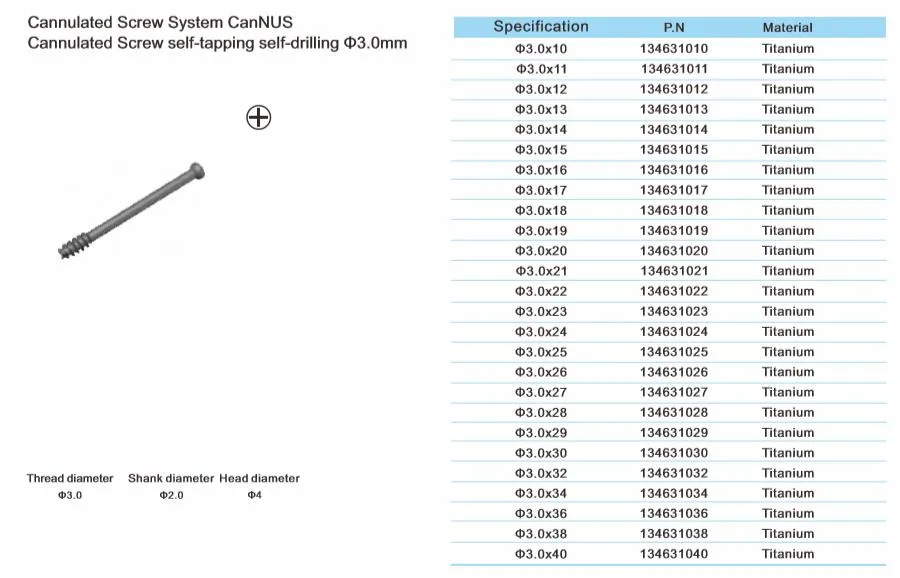
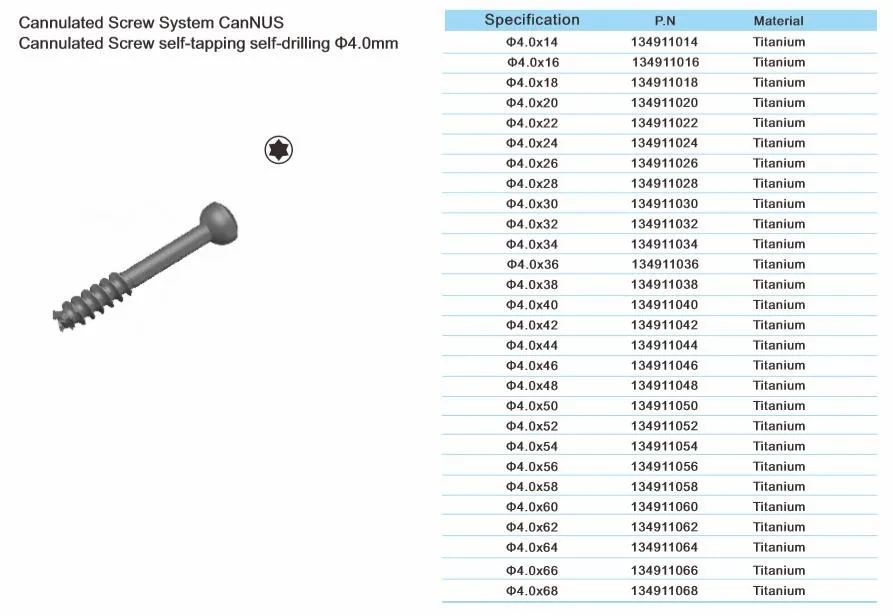
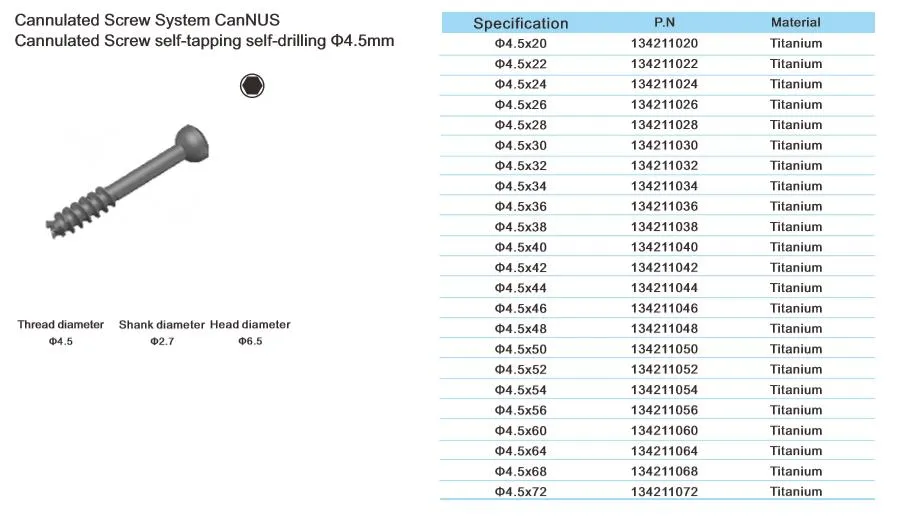
Cannulated Screw System CanNUS
Tornillos canulados
Cannulated Screw Φ6.5mm, self-drilling, thread length 32mm


Quality Assurance System
A complete Quality Assurance System has been established to continuously fulfill requirements according to ISO9001, 13485, QSR820, and CE, ranging from material procurement to design development and automated production processes.
The QMS consists of five major subsystems:
We continually improve the effectiveness of the quality management system to provide products and services that consistently meet and exceed expectations.
Frequently Asked Questions