Deep Dive: The Evolution of Orthopedic Instrument Manufacturing in Switzerland
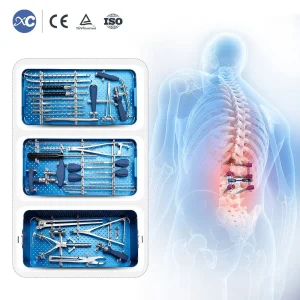
When discussing the Orthopedic Instrument Sets Manufacturer & Factory in Switzerland, one must understand the historical synergy between watchmaking and medical device engineering. The same machines used to create delicate gears for chronometers were adapted in the mid-20th century to create the first generation of high-precision surgical drills and screws. This heritage has evolved into a sophisticated ecosystem where biomedical engineering and metallurgy meet.
The Material Science Advantage
Swiss factories prioritize the use of high-grade Grade 5 Titanium (Ti6Al4V) and medical-grade stainless steel. These materials are processed through state-of-the-art CNC machining, ensuring that every 1.5mm micro fragment screw or 6.0mm spinal pedicle screw meets the exacting standards required for long-term implantation. The passivation and sterilization processes employed in Swiss-linked factories ensure zero-contaminant surfaces, which is critical for preventing post-operative infections in trauma surgery.
Innovative Applications in Spinal Surgery
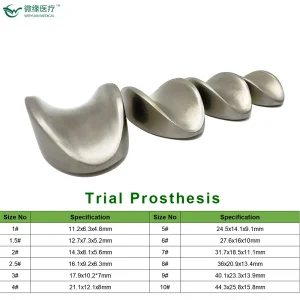
Spinal surgeries, such as posterior cervical fixation or lumbar rod installation, require instrument sets that offer both tactile feedback and mechanical reliability. Our Pedicle Screw Spinal Instrument Sets are designed with these needs in mind. The ergonomics of the handles and the torque-limiting drivers are optimized based on clinical feedback from European surgical teams, ensuring that the installation of titanium rods is as minimally invasive as possible.
The Growing Market for Veterinary Orthopedics in the EU
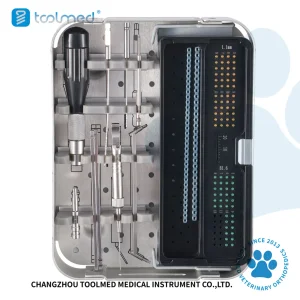
Switzerland serves as a gateway to the European veterinary market. As pet owners increasingly seek advanced medical care for their animals, the demand for specialized kits like the Advanced Locking Plate System (ALPS) has surged. These kits allow veterinarians to treat fractures in smaller animals with the same level of sophistication found in human hospitals. Our factory focuses on providing versatile kits (1.6mm to 4.0mm) that can adapt to the diverse needs of a modern veterinary practice, from urban Geneva to rural Alpine regions.
Sustainability and Manufacturing Efficiency
Modern Swiss industrial policy emphasizes Green Manufacturing. This involves reducing material waste during the milling process and utilizing renewable energy sources for factory operations. Our production lines are adopting these "Industry 4.0" practices, using AI-driven quality control to detect micro-defects that the human eye might miss. By integrating these technologies, we ensure that every instrument set shipped is a masterpiece of modern engineering.
The Role of ISO 13485 in Global Export
Compliance is the cornerstone of the Swiss medical industry. Adhering to the Medical Device Regulation (MDR) standards in Europe is mandatory. As a manufacturer aligned with these principles, Zhejiang Horaizon ensures that every step of the lifecycle—from the raw material sourcing to the final packaging—is documented and traceable. This transparency is why our orthopedic instrument sets are trusted by surgeons across borders.
 Horaizon Orthopedic Implants
Horaizon Orthopedic Implants