 Horaizon Orthopedic Implants
Horaizon Orthopedic Implants
 Horaizon Orthopedic Implants
Horaizon Orthopedic Implants
High-performance intramedullary systems designed for complex trauma recovery.
Norway represents one of the most sophisticated healthcare markets in the world, characterized by high standards of clinical excellence and a robust infrastructure for medical technology. As a leading Intramedullary Nails Manufacturer & Factory serving the Norwegian market, we recognize that the demand for high-quality orthopedic implants is driven by a combination of an aging population and a high incidence of trauma related to outdoor activities and extreme weather conditions.
Major hospitals in Oslo, Bergen, and Trondheim are at the forefront of adopting minimally invasive intramedullary nailing techniques for fracture fixation.
With a high prevalence of winter sports injuries (skiing, snowboarding), there is a specific need for implants that can withstand high-energy impacts.
The Norwegian Arthroplasty Register and trauma registries provide critical data that drive the iterative design of our intramedullary systems.
Orthopedic surgery in Norway has undergone a significant transformation over the last two decades. The shift from open reduction and internal fixation (ORIF) with plates to intramedullary (IM) nailing for long bone fractures is well-documented in Nordic clinical journals. Our factory specializes in producing nails that align with the Norwegian "biological fixation" philosophy, which prioritizes preserving the soft tissue envelope and optimizing blood supply to the fracture site.
Whether it is a femoral shaft fracture resulting from a high-speed traffic accident on the E6 highway or a fragility fracture in the elderly population of Tromsø, our intramedullary nails are engineered to provide immediate stability and facilitate early mobilization—a key metric for the Norwegian Healthcare System (Helse Sør-Øst, Helse Vest, etc.).
As a modern factory, we leverage AI-enhanced precision machining to ensure that every intramedullary nail meets the exacting tolerances required by Norwegian surgeons. Our manufacturing facility utilizes multi-axis CNC technology and robotic finishing to produce implants with superior surface textures. This is particularly important for the Norwegian market, where titanium alloys are preferred for their biocompatibility and MRI compatibility, essential for long-term patient monitoring in Norway's digitally advanced hospitals.
Our "Smart-Nail" initiative explores the integration of sensor technology to monitor bone healing progress, a trend gaining traction within the Oslo Medtech cluster. By focusing on data-driven design, we ensure that our products are not just pieces of metal, but intelligent medical solutions.
Norway has one of the highest hip fracture rates globally. Our Proximal Femoral Nail Antirotation (PFNA) systems are specifically designed for the osteoporotic bone often encountered in the elderly Norwegian population, providing superior cut-out resistance and stable fixation.
Trauma centers in Northern Norway frequently deal with complex fractures involving extreme cold conditions. Our titanium elastic nails and interlocking systems remain ductile and reliable, ensuring performance is never compromised by external environmental factors.
Furthermore, the decentralization of healthcare in Norway means that surgeons in smaller regional hospitals require instrumentation sets that are intuitive and comprehensive. Our intramedullary nail kits are designed for "first-time-right" surgery, reducing anesthesia time—a critical factor for patient safety in remote Norwegian municipalities.

Zhejiang Horaizon Medical Device Co., Ltd. is a company combined with development, manufacture and sales, and is specialized in orthopedic medical device.
Our company possesses advanced production equipment with high-precision and large-scale and complete sets of precision testing equipment. We have a professional team responsible for technical development. The company management is based on ISO9000 / ISO 13485 International Quality System Standard and YY/T0287 Medical Device Quality System Standard. We are always in pursuit of more stable and advanced product technology, and more convenient and simpler operational function.
Horaizon people adhere to the operation concept in mind, which is "precision, professional, innovative". We will strive to make new value for community and keep forging ahead for human health and happiness through excellent products with best quality and outstanding service!

Looking toward 2025 and beyond, the Norwegian orthopedic market is moving toward Personalized Medicine. As a forward-thinking manufacturer, we are integrating 3D modeling from CT scans to offer preoperative planning tools for complex intramedullary nailing procedures. This level of precision is increasingly demanded by specialized units like the Oslo University Hospital (Rikshospitalet).
Norway is a global leader in sustainability. Our factory is committed to green manufacturing processes, reducing waste in the production of titanium implants and utilizing recyclable packaging materials. We understand that Norwegian procurement agencies (Sykehusinnkjøp HF) place a high value on the environmental footprint of their suppliers. By choosing our intramedullary nails, Norwegian hospitals are partnering with a manufacturer that respects the Nordic commitment to the environment.
The biomechanical stability of an intramedullary nail is paramount. Our nails feature a unique anatomical curvature designed through extensive morphometric studies of European femur and tibia structures. For the Norwegian surgeon, this means easier insertion, reduced hoop stress, and a lower risk of iatrogenic fractures during the procedure. The interlocking screws are designed with a high-torque drive system to prevent stripping, ensuring that even in the toughest cortical bone, the fixation remains absolute.
Our interlocking nail systems are available in a variety of colors (Blue, Green, Gold) via anodic oxidation. This is not just for aesthetics; it aids in the rapid identification of nail types and sizes during high-pressure trauma surgeries in Norwegian ERs, enhancing surgical workflow efficiency.
Explore our full range of CE-certified implants for various anatomical locations.




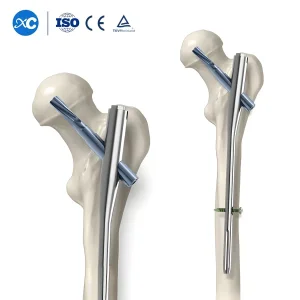


Choosing the right Intramedullary Nails Factory is critical for surgical success and patient satisfaction. With our deep understanding of the Norwegian medical landscape, commitment to ISO 13485 standards, and continuous innovation in material science, we are the preferred partner for hospitals and distributors across Norway. Our logistics network ensures timely delivery to even the most remote regions, supporting the Norwegian mission of equitable healthcare for all.